 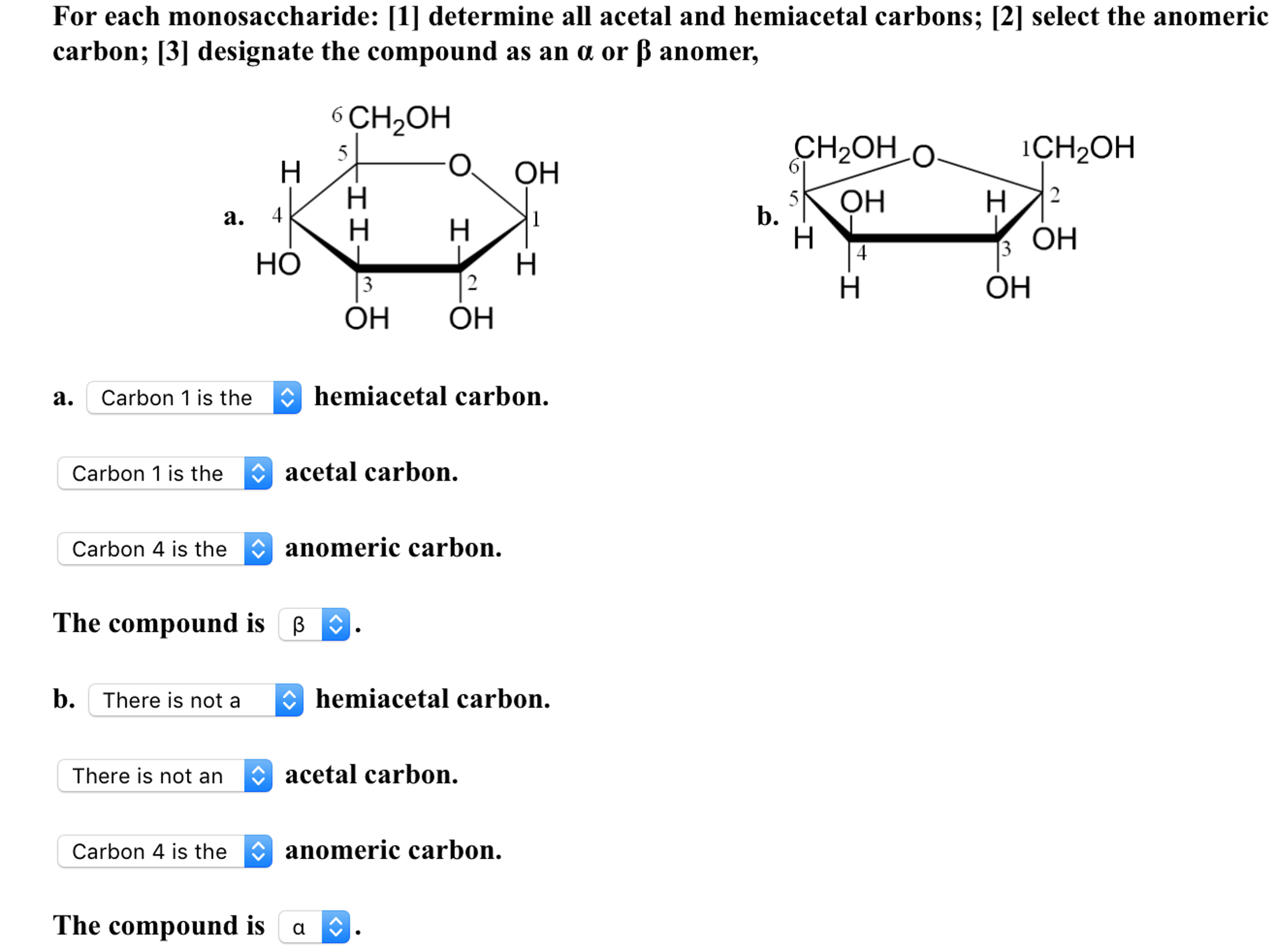 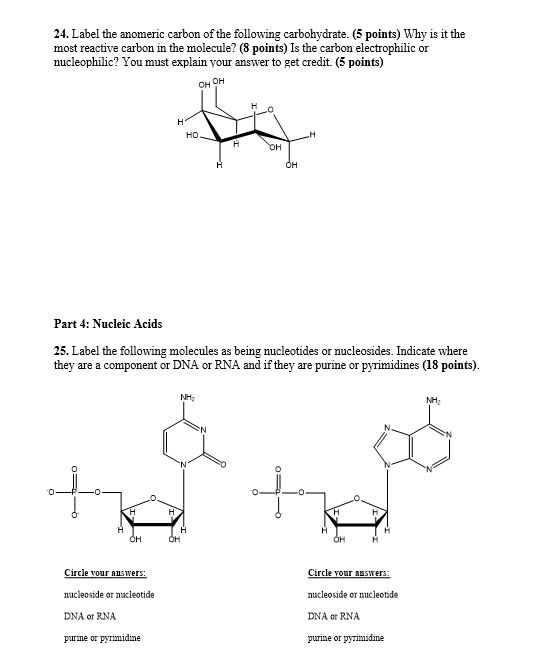
With knowledgeable experts and years of experience, Creative Proteomics offers accurate, reliable anomeric configuration analysis service.2312)/RD/Type/Annot/AP>endobj84 0 obj/ProcSet>/Type/XObject/BBox/FormType 1>streamĮndstreamendobj85 0 objendobj86 0 objendobj87 0 objendobj88 0 objendobj89 0 objendobj90 0 objstream The proton NMR spectrum also gives an idea about the constituent monosaccharides, based on chemical shifts and spin-spin coupling constants ( J H-H). In addition the α-anomeric proton resonates further down field (5.1 ppm) from the β–anomeric proton (4.5 ppm) making these two anomer distinguishable by 1H-NMR even at low field. 5-membered ring (4 carbons and 1 oxygen) ring closure occurs at carbon atom 2 (C2) C2 is the anomeric carbon atom in this molecule intermediate in. The signals arising due to anomeric protons usually appear in the range of 4.3~5.9 ppm, and also the protons of α-glycosides typically resonate 0.3-0.5 ppm downfield from those of the corresponding β–glycosides. The one-dimensional Proton Nuclear Magnetic Resonance Spectroscopy ( 1H-NMR) has been a source of valuable structural information of polysaccharides such as the number and the configuration of anomeric protons. The anomeric carbon is the carbon derived from the carbonyl carbon (the ketone or aldehyde functional group) of the open-chain form of the carbohydrate molecule. Example of 1H-NMR spectrum of α- and β–anomers Here, we report the synthesis of axial C1. There is also the influence of anomeric configuration on mechanochemical degradation of polysaccharides.įigure 2. Stereoselective reactions at the anomeric carbon constitute the cornerstone of preparative carbohydrate chemistry. For example, the anomeric effect is one of the major contributors to the stability of a certain anomer. They are different in structure, and thus have different stabilizing and destabilizing effects from each other. Polysaccharides with different anomeric configurations have different physical properties. Notice that all the initial chiral centers remain intact, and the two cyclic forms differ in the configuration of only one chiral center.We classify these as epimers, and in the case of sugars, they are said to be anomers.So, an anomer is a type of epimer characterized by the carbon in two possible configurations of a cyclic saccharide. Structural representation of D-glucose and its anomeric configurations These two forms are called anomers, and C 1 is called the anomeric carbon. These two forms are identical except for the configuration around C 1. There is the so-called mutarotation equilibrium between the α- and β-anomers of glucopyranose in aqueous solution.įigure 1. Depending on the orientation of C 1 when the C 5 hydroxyl bonds to it, two different forms can result. In this case the anomeric carbon atom is C-2, and the CH2OH is either above. 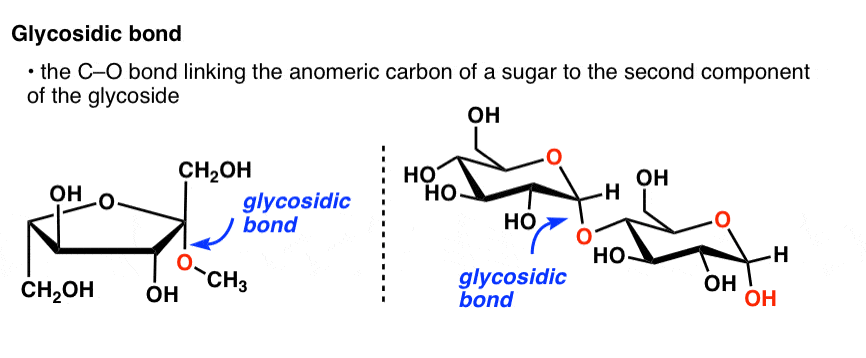
These two different stereoisomers (anomers) are designated α- and β-anomers. Carbon atoms are numbered beginning from the reactive end of the molecule. The two stereoisomers formed from the two possible stereochemistries at the anomeric center are called anomers. The anomeric center of a sugar is a stereocenter created from the intramolecular formation of an acetal (or ketal) of a sugar hydroxyl group and an aldehyde (or ketone) group. The new stereocenter is referred to as the anomeric carbon. Therefore, the formation of the cyclic structure from the open-chain structure is called anomerization. The signals corresponding to starch, sucrose, glucose, and fructose in. An anomeric carbon atom is derived from the carbonyl carbon of the open-chain structure of the carbohydrate. Download scientific diagram Anomeric carbon region of the spectra shown in Fig. They are different in the position of the attachment of a certain group to the new stereocenter. It is a type of epimer in which we can see an anomeric carbon atom. Cyclization causes the formation of two new diasteriomers. The anomeric carbon in a carbohydrate distinguishes hexoses from pentoses allows alpha/beta configurations as a new chiral center defines the molecule as D or L distinguishes pyranoses from furanoses Question 6 Match the carbohydrate to the structure name. During cyclization, the carbonyl moiety that formed part of that linear structure is transformed into a new stereocenter. the hemiacetal carbon of the cyclic form of a disaccharide Question: What is an anomeric carbon A. the acetal carbon of the cyclic form of a monosaccharide E. the hemiacetal carbon of the cyclic form of a monosaccharide D. Identification of the Anomeric ConfigurationĬarbohydrates usually exist in a linear or in a cyclic form, but given the stability, it is common that they are inclined to form rings. the acetal carbon of the cyclic form of a disaccharide C. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
